36. A Predictive Model of Acid Gas Rates of Absorption in Aqueous Amine Solvents
Keywords: absorption, acid gas, amine solvent, rate, model
36.1. Introduction
Acid gases in natural gas cause billions of dollars in damage each year. This application note illustrates the use of the MedeA materials modeling environment to optimize an industrial capture process, improve its efficiency, and reduce pollution. Acid gas capture can be achieved using several technologies. The most mature process deploys an absorption unit with an aqueous amine solvent [1] (Figure 36.1.1)
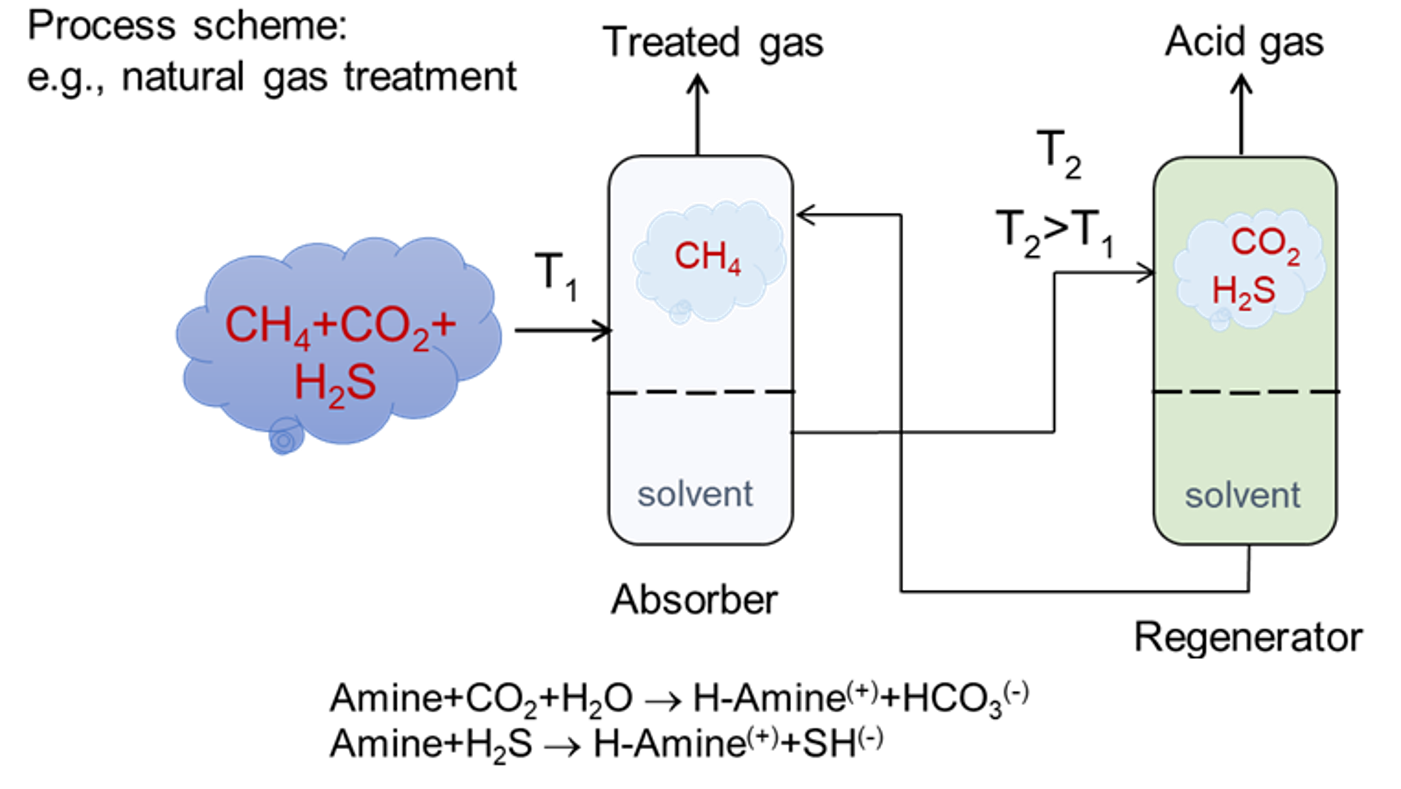
Figure 36.1.1 Schematic of an aqueous amine absorption unit.
The processes taking place in such an absorption unit are multi-physical and multi-scale [2]. In addition to the efficiency of the acid gas absorption unit, the regeneration unit is also a cause for concern, as it consumes energy to operate. In this application note, we focus on the initial absorption rates of H2S and CO2 in unsaturated aqueous amine solvents, for which good quality experimental rate values are available [3] (Figure 36.1.2).
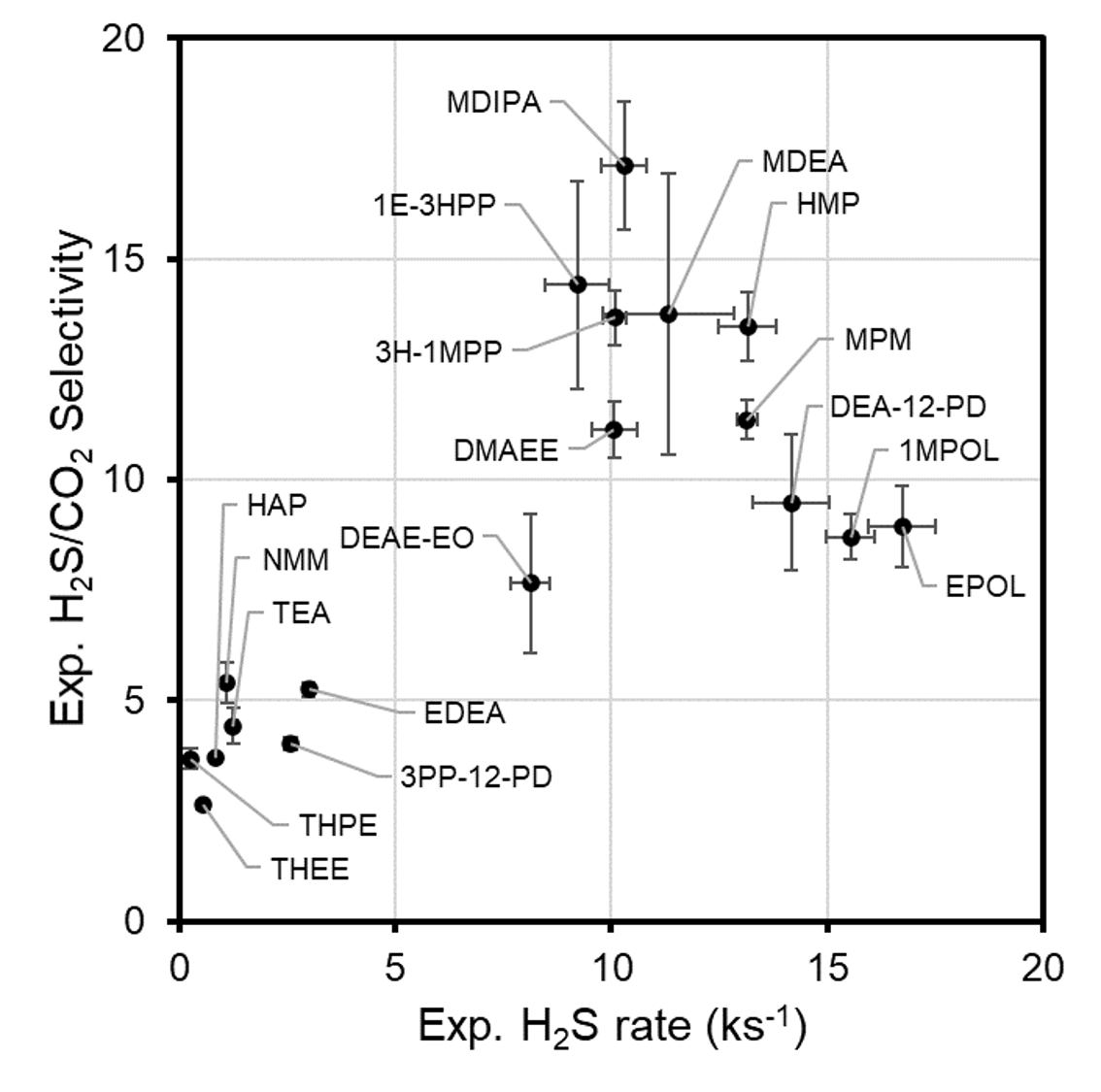
Figure 36.1.2 Experimental selectivity of absorption of H2S over CO2 as a function of the rate of absorption of H2S at T = 323.15 K in aqueous amine solvents (13 mol % amine, 87 mol % H2O) (Reprinted with permission from American Chemical Society).
This figure also shows the variation of the selectivity of the solvents for H2S. While CO2 is usually the major acid gas component (one order of magnitude higher), the tolerance threshold for H2S is much lower because of its toxicity. The aqueous amine solvent can in principle be tuned to precisely meet the chemical engineering requirements.
36.2. Results
Experimental absorption rate measurements are time-consuming, expensive, and further complicated by data quality requirements [3]. Therefore, relying on simulations is advantageous. However, one challenge remains: the conversion of the absorption rates to activation energies gives an energy scattering within 1.2 kJ mol-1 [3] [4]. This means that the accuracy required to obtain quantitative calculated data must be about 0.1 kJ mol-1. Such accuracy is beyond current quantum chemical methods [5]. In addition, the liquid phase system requires the sampling of many conformations to calculate accurate free energies [6].
A successful simulation protocol was developed to determine the rate-determining step in the absorption of H2S and CO2. It utilized the empirical Polanyi-Evans relationship [7] (Figure 36.2.1).
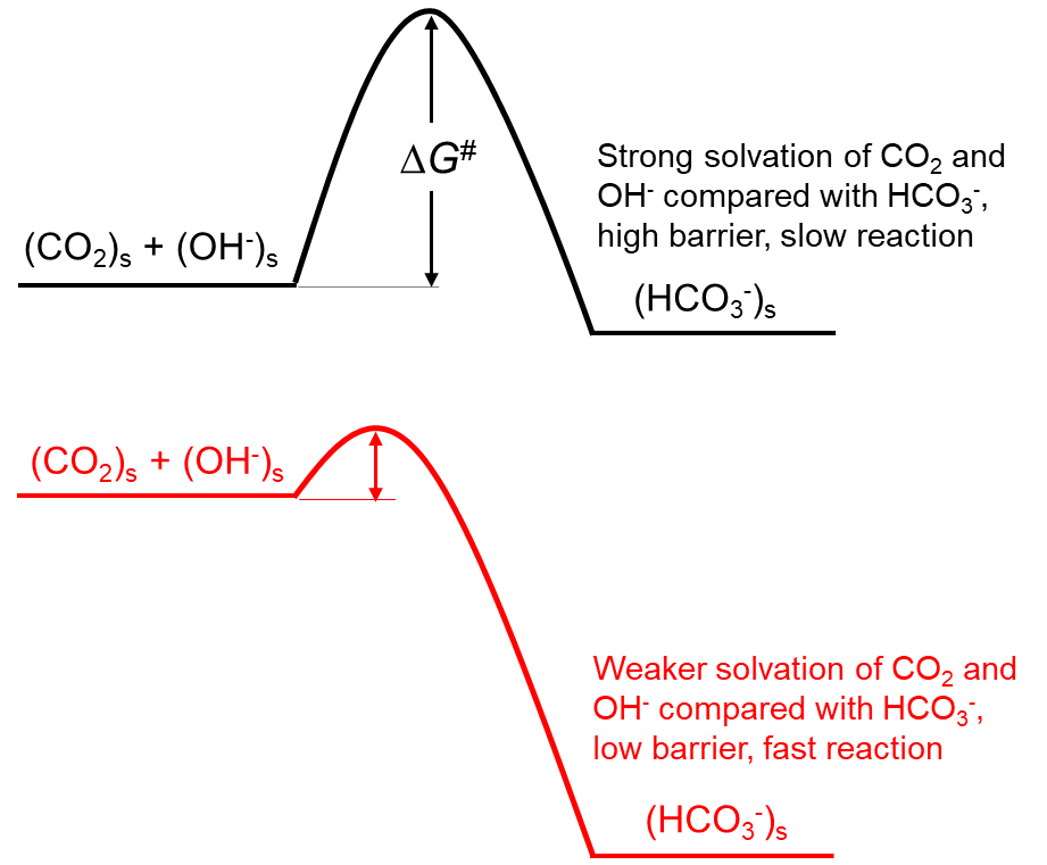
Figure 36.2.1 The Evans-Polanyi principle links linearly the variation of an activation energy barrier with changes in the solvation energies of the reactant and product (Reprinted with permission from American Chemical Society).
It allowed the estimation of activation energy from the energy difference between intermediates near the transition state, calculated using a classical (non-reactive) forcefield. This approach enabled extensive sampling of solvent and reaction intermediate conformations, far beyond what ab initio molecular dynamics and reactive forcefield simulations could achieve, resulting in highly accurate data. The protocol likely benefited from error compensation from the energy difference calculation, as the forcefield used, though not a latest generation forcefield, lead to good accuracy with a simple charge model of water (SPC model) and no polarizable charges.
The highly accurate calculated data combined with high-quality experimental absorption rate were used to fit the Polanyi-Evans relationship, leading to a quantitative prediction of the calculated absorption rates H2S [3] and CO2[4] compared to experimental rates (Figure 36.2.2).
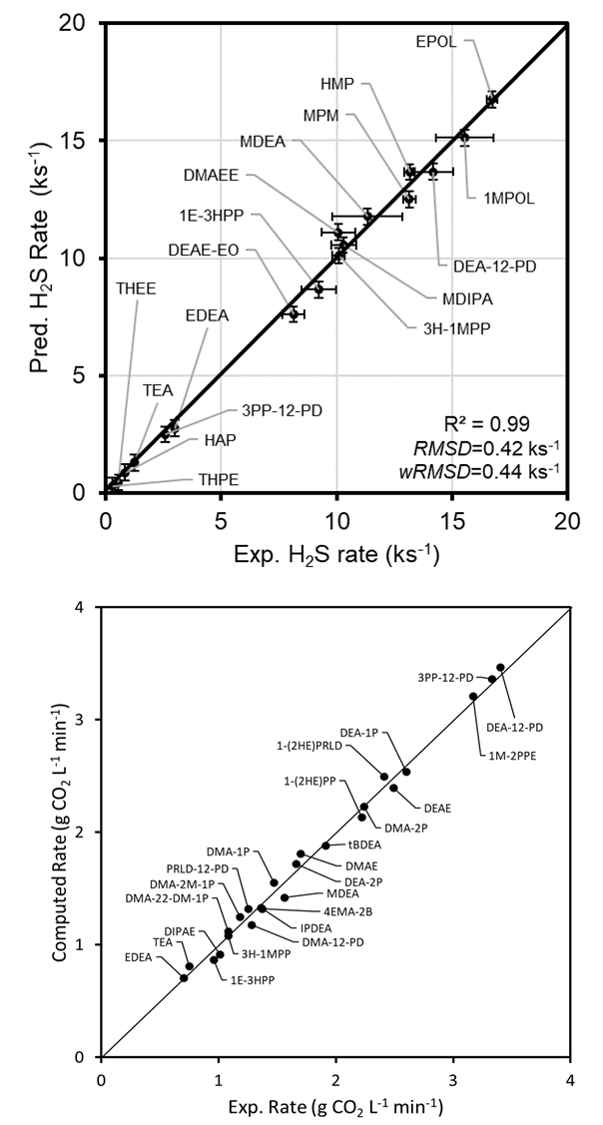
Figure 36.2.2 Experimental and computed rates of absorption of H2S (top) and CO2 (bottom) in aqueous amine solvents (Reprinted with permission from American Chemical Society).
The relative errors of the calculated CO2 and H2S absorption rates were 4% compared to the experimental ones.
36.3. Conclusions and Perspectives
The high accuracy of the calculated rates enabled the evaluation of a large number of solvents. This significantly guided experimental testing, reducing the time required by focusing on the most promising canditates. The calculations produced a substantial and consistent data set, facilitating the training of AI correlation models [3] [8]. These models were then used to predict the performance of over 10,000 aqueous amine solvents and their H2S/CO2 selectivity. In this application note, we focused on the absorption rate of acid gases in the absorption unit. A similar protocol could be applied to the study of the thermochemistry and kinetics of acid gas desorption, aiding in the optimization of energy consumption in the regeneration unit.
| [1] | F. de Meyer and S. Jouenne, “Industrial Carbon Capture by Absorption: Recent Advances and Path Forward.” Curr. Op. Chem. Eng. 38: 100868 (2022) (DOI). |
| [2] | M. Yiannourakou, X. Rozanska, B. Minisini, and F. de Meyer, “Molecular Simulations for Improved Process Modeling of an Acid Gas Removal Unit.” Fluid Phase Equil. 560: 113478 (2022). (DOI). |
| [3] | (1, 2, 3, 4, 5) X. Rozanska, A. Valtz, M. Riva, C. Coquelet, E. Wimmer, K. Gonzalez-Tovar, and F. de Meyer, “Selective H2S Absorption in Aqueous Tertiary Alkanolamine Solvents: Experimental Measurements and Quantitative Kinetic Model.” Ind. Eng. Chem. Res. 62(29): 11480-11490 (2023). (DOI). |
| [4] | (1, 2) X. Rozanska, E. Wimmer, and F. de Meyer, “Quantitative Kinetic Model of CO2 Absorption in Aqueous Tertiary Amine Solvents.” J. Chem. Inf. Model. 61(4): 1814-1824 (2021). (DOI). |
| [5] | M. Bursch, J. Mewes, A. Hansen, and S. Grimme, “Best-Practice DFT Protocols for Basic Molecular Computational Chemistry.” Angew. Chem. Int. Ed. 61: e202205735 (2022) (DOI). |
| [6] | M. P. Allen and D. J. Tildesley, “Computer Simulation of Liquids.” Oxford University Press (2017). |
| [7] | M. G. Evans and M. Polanyi, “Some Applications of the Transition State Method to the Calculation of Reaction Velocities, Especially in Solution.” Trans. Faraday Soc. 31: 875-894 (1935). (DOI). |
| [8] | A. A. Orlov, A. Valtz, C. Coquelet, X. Rozanska, E. Wimmer, G. Marcou, D. Horvath, B. Poulain, A. Varnek, and F. de Meyer, “Computational Screening Methodology Identifies Effective Solvents for CO2 capture.” Comm. Chem. 5(1): 37 (2023). (DOI). |
| download: | pdf |
|---|